555-555-5555
mymail@mailservice.com
Kelulusan
Adakah ubat ini diluluskan?
Jawapannya ialah YA. Ubat ini diluluskan untuk digunakan di Amerika pada tahun 2000. Ubat ini telah wujud 22 tahun di pasaran. Ubat ini diluluskan oleh FDA iaitu badan pemerhati pharmasi terbesar di dunia. FDA di asaskan di Amerika Syarikat pada Jun 1906 antara yang tertua di dunia jika dibandingkan dengan KKM. FDA ini juga yang meluluskan vaccine pfizer, moderna dan sinovac. Oleh itu anda tidak perlu risau , kerana ubat ini di jamin selamat.
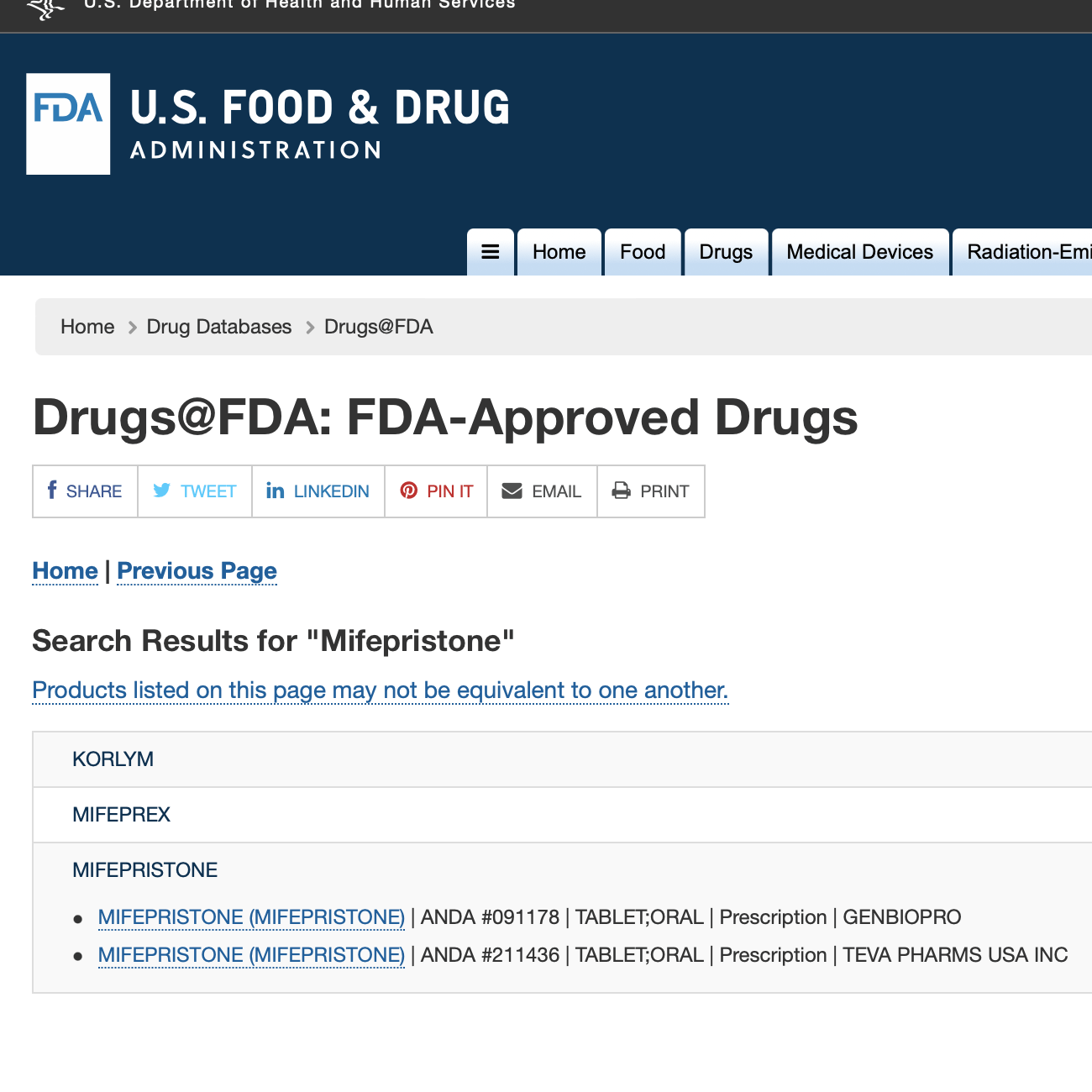
Mifepristone
Mifeprex (mifepristone) is used, together with another medication called misoprostol, to end an early pregnancy. FDA first approved Mifeprex in 2000. In 2016, the agency approved a supplemental application for Mifeprex based on data and information submitted by the drug manufacturer. After reviewing the supplemental application, the agency determined that Mifeprex is safe and effective when used to terminate a pregnancy in accordance with the revised labeling. In 2019, FDA approved a generic version of Mifeprex, Mifepristone Tablets, 200 mg.
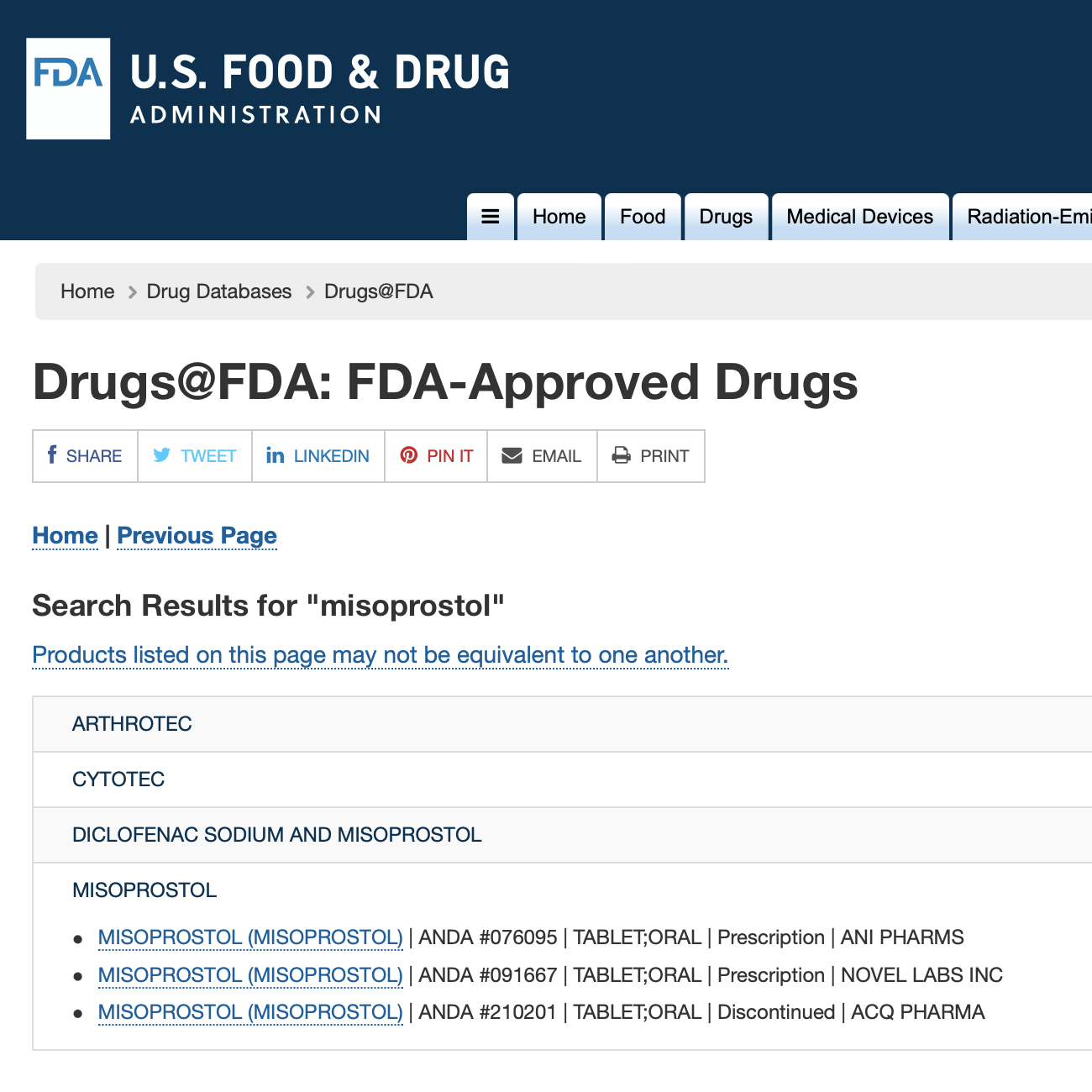
Misoprostol
Misoprotol is approved, in a regimen with misoprostol, to end a pregnancy through 70 days gestation (70 days or less since the first day of a woman’s last menstrual period).
MTP Kit
FDA previously approved a REMS for Mifeprex. In 2019, at the same time FDA approved the generic version of Mifeprex, the agency approved a single, shared system REMS for mifepristone products for the medical termination of intrauterine pregnancy through 70 days gestation (the Mifepristone REMS Program). Under the 2019 REMS
Lokasi
Kuala Lumpur
Johor Bharu
Penang
Melaka
+60196016677
ubatwanita@gmail.com
Subscribe to our newsletter
We will get back to you as soon as possible.
Please try again later.